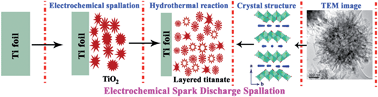
An ultrafast and template-free method to synthesize three-dimensional (3D) hierarchical layered titanate microspherulite (TMS) particles with high surface area is reported. The synthesis makes use of an electrochemical spark discharge spallation (ESDS) process, during which a fast anodic reaction on the titanium surface creates a layer of titanium dioxide that instantly breaks down by the applied electrical field into the solution in the form of titanium oxide particles. The spalled particles readily react with the heated NaOH electrolyte to form the titanate particles. A typical as-prepared TMS with a diameter of 0.4∼1.5 μm is synthesized by ESDS of Ti foils in 10 M NaOH solution under an applied current density of 0.5 A cm−2, leading to a reaction yield of approximately 0.10∼0.15 g per square centimetre of exposed Ti foil within 20 min. After hydrogen ion exchange, the surface area can reach as high as ∼406 m2 g−1. On the Ti surface, a crystalline rutile TiO2 nanosheet structure is formed, which is attributed to the local exothermic heat caused by the spark discharge. A formation mechanism of the TMS is discussed based on field emission scanning electron microscopy (FESEM), a transmission electron microscopy (TEM) study and Raman scattering spectroscopy analysis. The as-prepared TMS shows excellent adsorption performance compared with a titanate micro-particle (TMP), nanowire (TNW) and nanotube (TNT) when methylene blue (MB) and PbII ions are used as representative organic and inorganic pollutants. The mechanism of adsorption has also been discussed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...