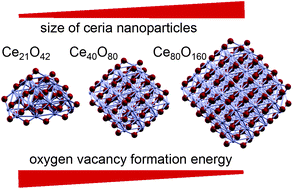
We address the formation of the energetically most favourable single oxygen vacancies in ceria nanoparticles (CeO2)n focusing on their size dependence. We study a series of structures with increasing number of CeO2 units (n = 21, 30, 40 and 80) that, according to well tested interatomic-potential calculations, approach the global minima for these particle sizes. The structures thus obtained are refined by means of density functional (DF) methods, modified by the on-site Coulomb correction. Subsequent DF calculations are performed to quantify and analyse the depletion of atomic O from the nanoparticles that results in the formation of a vacancy Ovac. We show that (i) removal of a low- (two-)coordinate O atom from ceria species requires the lowest energy, in line with evidence from other metal oxides; (ii) the depletion of such O atoms from the nanoparticles is strongly facilitated compared to extended (even irregular) surfaces; (iii) increase of the particle size is accompanied by a dramatic decrease of the Ovac formation energy, implying that at certain sizes this energy should reach a minimum; (iv) the size dependence of the Ovac formation energy is driven by the electrostatics, thus enabling the prediction of the most easily removable O atoms by analysing the distribution of the electrostatic potential in the pristine stoichiometric (vacancy-free) ceria systems. Our findings provide a key to rationalize the observed spectacularly enhanced reactivity of ceria nanostructures.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...