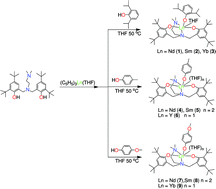
A series of neutral lanthanide aryloxides supported by an amine bridged bis(phenolate) ligand were synthesized, and their catalytic behavior for the polymerization of L-lactide was explored. The reactions of (C5H5)3Ln(THF) with amine bridged bis(phenol)LH2 [L = Me2NCH2CH2N{CH2-(2-O-C6H2-But2-3,5)}2] in a 1 : 1 molar ratio, and then with 1 equivalent of 2,6-diisopropylphenol, p-cresol or 4-methoxyphenol, respectively, in situ in THF gave the neutral lanthanide aryloxides LLn(OC6H3-2,6-Pri2)(THF) [Ln = Nd (1), Sm (2), Yb (3)], LLn(OC6H4-4-CH3)(THF)n [Ln = Nd (4), Sm (5), n = 2; Ln = Y (6), n = 1] and LLn(OC6H4-4-OCH3)(THF)n [Ln = Nd (7), Sm (8), n = 2; Ln = Yb (9), n = 1] in high isolated yields. These complexes were well characterized by elemental analyses, IR spectra and NMR spectroscopy in the case of complex 6. The definitive molecular structures of complexes 1–8 were determined by single-crystal X-ray analyses, which revealed that both the substituents of the aryloxo groups and the ionic radii of the lanthanide metals affect the solid-state structures of the bis(phenolate) lanthanide aryloxides. It was found that complexes 1–9 are efficient initiators for the ring-opening polymerization of L-lactide, and the structures of the aryloxo groups have no obvious effect on the catalytic activity and controllability. A further study revealed that complex 6 can initiate the highly heteroselective ring-opening polymerization of rac-lactide.

 Please wait while we load your content...
Please wait while we load your content...