Oxovanadium(iv) complexes of phenanthroline bases: the dipyridophenazine complex as a near-IR photocytotoxic agent†
Abstract
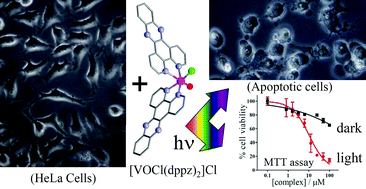
Oxovanadium(IV) complexes [VOCl(B)2]Cl (1–3) of phenanthroline bases (B), viz. 1,10-phenanthroline (phen in 1), dipyrido[3,2-d:2′,3′-f]quinoxaline (dpq in 2) and dipyrido[3,2-a:2′,3′-c]phenazine (dppz in 3), have been prepared, characterized and their DNA and protein binding, photo-induced DNA and protein cleavage activity and photocytotoxicity have been studied. Complex 2, structurally characterized by X-ray crystallography, shows the presence of a vanadyl group in VOClN4 coordination geometry. The dpq ligand displays a chelating mode of binding with a N-donor site trans to the oxo-group. The chloride ligand is cis to the oxo-group. The one-electron paramagnetic complexes show a d–d band near 715 nm in 15% DMF-Tris-HCl buffer. The complexes are redox active exhibiting a V(IV)/V(III) redox couple within −0.5 to −0.7 V vs. SCE in 20% DMF-Tris-HCl/0.1 M KCl. The complexes bind to calf thymus (CT) DNA in the order: 3 (dppz) > 2 (dpq) > 1 (phen). The binding data reveal the groove and/or partial intercalative DNA binding nature of the complexes. The complexes show “chemical nuclease” activity in the dark in the presence of 3-mercaptopropionic acid or hydrogen peroxide via a hydroxyl radical pathway. The dpq and dppz complexes are efficient photocleavers of DNA in UV-A light of 365 nm forming reactive singlet oxygen (1O2) and hydroxyl radical (˙OH) species. Complexes 2 and 3 also show DNA cleavage activity in red light (>750 nm) by an exclusive ˙OH pathway. The complexes display a binding propensity to bovine serum albumin (BSA) protein giving KBSA values in the range of 7.1 × 104–1.8 × 105 M−1. The dppz complex 3 shows BSA and lysozyme protein cleavage activity in UV-A light of 365 nm via˙OH pathway. The dppz complex 3 exhibits significant PDT effect in human cervical cancer HeLa cells giving IC50 values of 1.0 μM and 12.0 μM in UV-A and visible light, respectively (IC50 = >100 μM in the dark).

 Please wait while we load your content...
Please wait while we load your content...