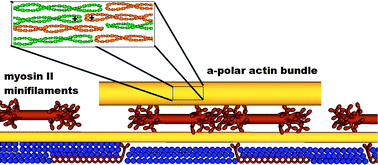
The cooperative action of many molecular motors is essential for dynamic processes such as cell motility and mitosis. This action can be studied by using motility assays in which the motion of cytoskeletal filaments over a surface coated with motor proteins is tracked. In previous studies of actin-myosin II systems, fast directional motion was observed, reflecting the tendency of myosin II motors to propagate unidirectionally along actin filaments. Here, we present a motility assay with actin bundles consisting of short filamentous segments with randomly alternating polarities. These actin tracks exhibit bidirectional motion with macroscopically large time intervals (of the order of several seconds) between direction reversals. Analysis of this bidirectional motion reveals that the characteristic reversal time, τrev, does not depend on the size of the moving bundle or on the number of motors, N. This observation contradicts previous theoretical calculations based on a two-state ratchet model (M. Badoual, F. Jülicher and J. Prost, Proc. Natl. Acad. Sci. USA, 2002, 99, 6696), predicting an exponential increase of τrev with N. We present a modified version of this model that takes into account the elastic energy due to the stretching of the actin track by the myosin II motors. The new model yields a very good quantitative agreement with the experimental results.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...