Recognition properties of receptors based on dimesitylmethane-derived core: Di- vs.monosaccharide preference†
Abstract
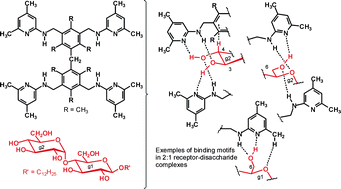
Dimesitylmethane-derived receptors 12 and 13, incorporating four heterocyclic recognition groups capable of serving as hydrogen bonding sites, were designed to recognize

 Please wait while we load your content...
Please wait while we load your content...