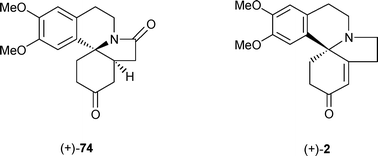
A concise asymmetric total synthesis of (+)-erysotramidine is described, using chiral base desymmetrisation of a meso-imide, N-acyliminium addition, retro-Diels–Alder cycloaddition and radical cyclisation as the key steps. A related route, starting from a cyclobutene-fused imide, was explored, and established a novel construction of the Erythrina alkaloid skeleton using a key ring-opening/ring-closing metathesis step. Completion of this synthesis was thwarted by problems with the removal of an unwanted vinylic side-chain. Complementary enantiospecific routes to Erythrina systems were explored, starting from (L)-malic acid. Some unexpected observations were made concerning the diastereocontrol in malic acid-derived N-acyliminium ion cyclisations, where changing the protecting group of the alcohol function from acetate to OTIPS resulted in a dramatic change in diastereocontrol. Products from these reactions could be transformed into known intermediates for natural alkaloids, and into (+)-demethoxyerythratidinone itself, by means of radical cyclisations or intramolecular aldol reactions as the key steps.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...