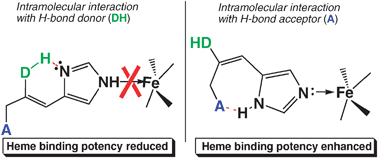
Intramolecular hydrogen bonding as a determinant of the inhibitory potency of N-unsubstituted imidazole derivatives towards mammalian hemoproteins†
Abstract
Enzymes involved in the mammalian microsomal
We are excited to let you know that our journals content will be migrating to the Silverchair platform, with a planned launch in summer 2026.
All of our content will still be hosted at pubs.rsc.org and the new platform will provide a more intuitive reading and navigation experience, along with improved discovery and indexing of your work.
Details on the move can be found in our partnership announcement.
* Corresponding authors
a
URA 2096 du CNRS, Institut de Biologie et Technologies de Saclay, Commissariat à l’Energie Atomique, Bâtiment 528, CEA-Saclay, Gif-sur-Yvette, France
E-mail:
marcel.delaforge@cea.fr
Fax: +33 1 69 08 87 17
Tel: +33 1 69 08 68 39
b INSERM U620; Université de Rennes 1, IFR 140, 2 Avenue du Pr. Léon Bernard, Rennes, France
c
Laboratoire de Chimie Bioorganique et Bioinorganique, CNRS, Institut de Chimie Moléculaire et des Matériaux d’Orsay, UMR 8182, Bâtiment 420, Université Paris-Sud XI, Orsay, France
E-mail:
jpmahy@icmo.u-psud.fr
Fax: +33 1 69 15 72 81
Tel: +33 1 69 15 74 21
d Department of Chemistry, University of Pennsylvania, 231 South 34th Street, Philadelphia, USA
Enzymes involved in the mammalian microsomal

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
