Controlled deposition of cells in sealed microfluidics using flow velocity boundaries
Abstract
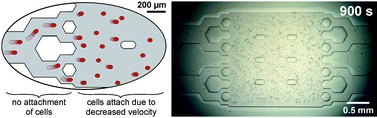
We present a method for depositing cells in a sealed microfluidic device. The device consists of a poly(dimethylsiloxane) (PDMS) microfluidic network (MFN) sealed with a Si chip. The Si chip has vias and ports that are connected to high-precision motorized pumps. The surfaces of the PDMS MFN are homogeneously coated with fibronectin cell adhesion molecules (CAMs). Flow velocity boundaries are created between vicinal microfluidic structures to prevent or permit deposition of cells in specific regions of the MFN. In narrow flow paths, cells experience a wall shear stress from the fast-moving liquid that overcomes the initial adhesion of the cells with CAMs. Conversely, cells can adhere to CAMs in larger flow paths such as cell chambers inside which the velocity of the liquid and the shear stress are reduced. Interactively changing pumping rates makes the critical velocity (the velocity at which cells deposit in the chamber but not elsewhere) easy to find. The transparent PDMS MFN allows both real-time visualization of the deposition process and cellular assays. We illustrate this method using N9 mouse microglia cells. In one experiment, ∼75 microglia are deposited per min in a ∼0.5 µL chamber. The deposited cells remain viable, as assessed from staining and biofunctional assays. This method is simple, reliable, fast, and flexible, and therefore is an attractive technique for depositing cells in microfluidic systems for numerous applications.

 Please wait while we load your content...
Please wait while we load your content...