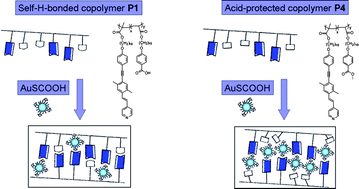
A series of photoluminescent (PL) and liquid crystalline (LC) self-H-bonded side-chain copolymers (P1–P3) consisting of pyridyl H-acceptors and isomeric acid H-donors (i.e., para-, meta-, and ortho-benzoic acids) were synthesized. Supramolecular H-bonded complexes were also obtained by mixing the photoluminescent H-acceptor homopolymer PBT1 (containing pyridyl pendants) with isomeric H-donor homopolymers P7–P9. The formation of H-bonds was confirmed by FTIR, DSC, and XRD measurements. Moreover, PL and LC properties of the H-bonded copolymers and complexes were affected not only by the H-bonding effect of the supramolecular structures but also by the acid-substituted positions of isomeric H-donors. In combination with different functionalized gold nanoparticles (which bear acid or acid-free surfactants), the emission intensities of nanocomposites containing self-H-bonded copolymer P1 (bearing both H-acceptor and H-donor moieties) and non-self-H-bonded copolymer P4 (bearing acid-protected moieties), were quenched to different extents by varying the concentration of gold nanoparticles. The copolymeric H-acceptors and surface-modified gold nanoparticles demonstrated diverse morphological and PL quenching effects on the supramolecular architectures of the nanocomposites, which result from competition between the H-donors from the acid pendants on copolymers and the acid surfactants on the gold nanoparticles.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...