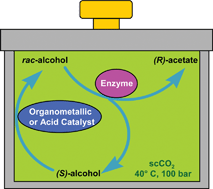
The kinetic resolution (KR) and dynamic kinetic resolution (DKR) of racemic 1-phenylethanol with the acyl donor vinyl acetate catalysed by Pseudomonas cepacia lipase ATCC 21808 (PCL) (this has recently been reclassified as Burholderia cepacialipase), either as the suspended native protein (PCL) or immobilised on ceramic particles (commercial lipase Amano PS CI) has been examined, as batch reactions, in supercritical carbon dioxide (scCO2). The yields and enantioselectivities achieved in these reactions were comparable to experiments conducted in hexane. The immobilised lipase PS CI displayed excellent yields (48–49%) and enantioselectivities (98–99%) to give (R)-phenylethyl acetate after 2.5 hours reaction at 40 °C in scCO2. The acylated product was obtained in higher yield in scCO2 compared to the reaction carried out in hexane (yield 30–35%; ee 98–99%) under comparable conditions. Unlike PCL, the immobilised lipase PS CI could then be successfully reused with no loss of activity or selectivity observed over four reaction cycles. Combining lipase PS CI with a chemical catalyst (either the metal catalyst [Ru(p-cymene)Cl2]2 or the acid catalyst Nafion SAC 13) to catalyse the racemisation of the unreacted alcohol caused an increase in the yield of the (R)-acyl phenylethanol product to 70 and 85%, respectively, in scCO2 indicating a viable dynamic kinetic resolution system had been established. The enantioselectivities of the products in these reactions were slightly higher in scCO2 (96% Ru-catalyst; 85% Nafion) compared to those observed in hexane (91%; 81%). The lower enantioselectivity with the acidic Nafion SAC 13 catalyses indicates that some non-enzymic acid catalysed esterification was occurring.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...