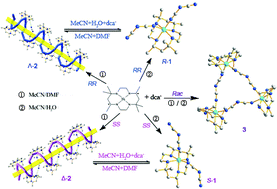
The reactions of [Ni(α-SS-L)](ClO4)2/[Ni(α-RR-L)](ClO4)2 with dca− gave two enantiomers of [Ni(SS-L)](dca)2 (S-1) and [Ni(RR-L)](dca)2 (R-1) in acetonitrile/water, and two supramolecular stereoisomers of {[Ni(SS-L)]2(dca)3(ClO4)·DMF}n (Δ-2) and {[Ni(RR-L)]2(dca)3(ClO4)·DMF}n (Λ-2) in acetonitrile/DMF, respectively (L = 5,5,7,12,12, 14-hexamethyl-1,4,8,11-tetraazacyclotetradecane, dca = dicyanamide, N(CN)2−). While the reaction of racemic [Ni(α-rac-L)](ClO4)2 (containing an equal amount of SS and RR enantiomers) with dca− in acetonitrile/water or acetonitrile/DMF produced a trimer of [Ni(rac-L)(dca)]3·(dca)·(ClO4)2·MeCN (3) rather than S-1/R-1 or Δ-2/Λ-2. X-ray single-crystal analysis reveals that S-1/R-1 and Δ-2/Λ-2 crystallize in a chiral space groupP21, while 3 crystallizes in a centrosymmetric space groupC2/c. In S-1/R-1, the [Ni(SS-L)](dca)2/[Ni(RR-L)](dca)2 monomers are linked through intermolecular hydrogen bonds to form a two-dimensional sheet. While the intermolecular hydrogen bonding linking of {[Ni(SS-L)]2(dca)3}+/{[Ni(RR-L)]2(dca)3}+ leads to the formation of a 1D right-handed helical chain of Δ-2 and 1D left-handed helical chain of Λ-2, respectively, demonstrating there is a correlation between the helicity of 1D chains and the chirality of building blocks. Complex 3 forms a triangular structure of {[Ni(rac-L)(dca)]3}3+, in which the macrocyclic ligand L adopts unsymmetrical RR/RR/SS or RR/SS/SS configurations. The homochiral nature of S-1/R-1 and Δ-2/Λ-2 are confirmed by the results of solid circular dichroism (CD) spectra measurements. The magnetic properties of chiral Δ-2/Λ-2 and achiral 3 were investigated.

 Please wait while we load your content...
Please wait while we load your content...