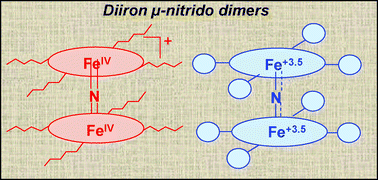
μ-Nitrido-bis [tetra-(hexyl-sulfonyl)phthalocyaninatoiron] (3a) and μ-nitrido-bis [tetra-(tert-butylsulfonyl) phthalocyaninatoiron] (3b) complexes have been prepared and fully characterized by electrospray ionization mass spectrometry, UV-Vis, FTIR, EPR, Mössbauer techniques as well as by X-ray photoelectron and Fe K-edge X-ray absorption spectroscopies. Small changes at the periphery of the phthalocyanine ligand introduce a difference in the iron oxidation state. While 3b with tert-butyl substituents is a neutral complex with a mixed-valence Fe3.5–N–Fe3.5 structural unit, 3a having n-hexyl substituents is an oxidized cationic FeIV–N–FeIV complex. The structural parameters of N-bridged diiron phthalocyanine with a Fe3.5–N–Fe3.5 unit were determined for the first time. Iron atoms in 3b are displaced out of plane by 0.24 Å and the Fe–N bond distance of the linear Fe–N–Fe fragment is equal to 1.67 Å. Both complexes selectively catalyze benzylic oxidation of alkyl aromatic compounds by tBuOOH. Toluene was oxidized to benzoic acid with 80% selectivity, and the total turnover number was as high as 197. p-Toluic acid was the principal product of p-xylene oxidation. In this case the turnover number achieved 587 substrate molecules per molecule of catalyst. The described catalytic system is complementary to the recently reported system based on μ-nitrido diiron tetrabutylphthalocyanine–H2O2 which effectively oxidizes the benzene ring.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...