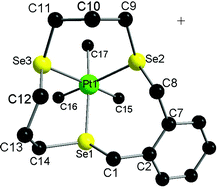
Simultaneous dropwise addition of thf/EtOH solutions of Se{(CH2)3OTs}2 and o-C6H4(CH2SeCN)2 or NCSe(CH2)3SeCN to a suspension of NaBH4 in thf/EtOH at room temperature yields gram quantities of the 13- and 12-membered triselenoether macrocycles L1 and L2 respectively in high yield. The 11-membered ring L3 is obtained similarly by simultaneous dropwise addition of thf/EtOH solutions of Na2[o-C6H4Se2] (itself prepared by NaBH4reduction of the polymeric [o-C6H4Se2]n) and Se{(CH2)3OTs}2 to a suspension of NaBH4 in thf/EtOH. The small-ring, potentially tridentate Se2N(pyridyl)-donor macrocycles L4 and L5 were obtained in essentially quantitative yield by simultaneous dropwise addition of thf/EtOH solutions of 2,6-bis(bromomethyl)pyridine and either o-C6H4(CH2SeCN)2 or NCSe(CH2)3SeCN to a suspension of NaBH4 in thf/EtOH at room temperature. L1–L5 have been characterised by 1H, 13C{1H} and 77Se{1H} NMR spectroscopy, EI MS, and for L1 and L4, by X-ray crystal structures. Reaction of PtMe3I with one mol. equiv. of L (L = L1–L5) in refluxing CHCl3 gives the ionic complexes [PtMe3(L)]I cleanly and in good yield. These were characterised by 1H, 13C{1H}, 77Se{1H} and 195Pt NMR spectroscopy, electrospray MS, microanalyses and by crystal structures of [PtMe3(L1)]I and [PtMe3(L4)]I, which confirm distorted octahedral coordination at Pt(IV), with fac-tridentate coordination of the macrocycle in all cases, with anionic iodide. The complexes [PtCl2(L)] (L = L1–L3) were obtained as poorly soluble yellow-orange solids by reaction of PtCl2 with L in MeCN solution. The d3Cr(III) complexes of L (L = L1–L5) were obtained by reaction with [CrCl3(thf)3] in anhydrous CH2Cl2 as distorted octahedral fac-[CrCl3(L)], as poorly soluble blue-purple through to green powdered solids, which have been characterised by microanalysis, UV-visible and IR spectroscopy and by their magnetic moments. The properties of these complexes are compared with related chalcogenoether complexes from the literature involving thioether and acyclic selenoether coordination.

 Please wait while we load your content...
Please wait while we load your content...