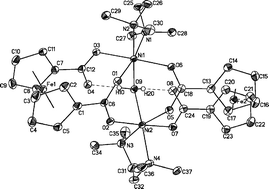
The synthesis and characterisation of a series of novel complexes of type [((tmeda)M-OC(O)-fc-(μ-CO2))2(μ-H2O)] (M = Co, 4; M = Ni, 5), zwitter-ionic [((pmdta)(H2O)Cu+-OC(O)-fc-CO2−)(CH3OH)] (9), and coordination polymer [(tmeda)Cu((OC(O))2fc)]n (7) (fc = ferrocene-1,1′-diyl, (η5-C5H4)2Fe; tmeda = N,N,N′,N′-tetramethylethylenediamine; pmdta = 1,1,4,7,7-N,N,N′,N″,N″-pentamethyldiethylenetriamine) composed of ferrocene dicarboxylates and [(tmeda)/(pmdta)M]2+ entities is reported. These complexes could be prepared from [M(NO3)2(tmeda)] (M = Co, 2; M = Ni, 3; M = Cu, 6) and [Cu(NO3)2(pmdta)] (8), respectively, with stoichiometric amounts of 1,1′-ferrocene dicarboxylic acid (1) in presence of [n-Bu4N]OH–H2O. The molecular structures of 4, 5, 7, and 9 in the solid state have been determined by single-crystal X-ray structure analysis. Electrochemical and UV-vis–NIR spectroscopic studies of these complexes are discussed in terms of electronic communication between the redox-active transition metals. In addition, the magnetic properties of 4 and 5 have been studied by susceptibility measurements vs. temperature. In complex 4, the two cobalt(II) ions are antiferromagnetically coupled, while in 5 the Ni–Ni coupling is ferromagnetic with J≈ +2.2(1) cm−1.

 Please wait while we load your content...
Please wait while we load your content...