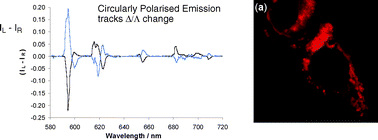
Six pairs of Eu(III) and Tb(III) complexes of macrocyclic ligands, incorporating a common tetraazatriphenylene sensitiser, have been examined in terms of their solution structure, sensitivity to excited state quenching, protein affinity, cell toxicity and preliminary cell localisation profiles. A complex with three (S)-phenylalanine-derived ligating groups possesses distinctive 1H NMR, Eu emission and circularly polarised emission properties, consistent with a unique Λ-configuration in the 9-coordinate complex, where an amide carbonyl group occupies the capping position of the coordination polyhedron. Each complex possesses similar sensitivity to quenching by ascorbate, urate and iodide, has similar toxicity behaviour and shows a common intracellular localisation profile that is consistent with compartmentalisation in lysosomes or late endosomes. Such behaviour accords with the hypothesis that it is the nature of the sensitising moiety that determines each of these properties.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...