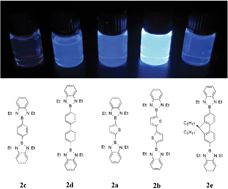
A series of π-conjugated systems with two and three 1,3-diethyl-1,3,2-benzodiazaborolyl end-groups was synthesised in 58–91% yields using established 1,3,2-diazaborole methodologies. The bis(diazaborolyl) compounds contain thiophene -2,5-C4H2S- (2a), dithiophene -5,5′-(2,2′-C4H2S)2- (2b), phenylene -1,4-C6H4- (2c), biphenylene -4,4′-(1,1′-(C6H4)2)- (2d) and dioctylfluorene -2,7-(9,9-(C8H7)2C11H6)- (2e) bridges. The three-way linkers in the tris(diazaborolyl) assemblies contain a central phenylene unit -1,3,5-C6H3- linked to the borolyl end groupsviathiophene -2,5-C4H2S- (3a), directly bonded (3b) or viaphenylene -1,4-C6H4- (3c) units. Molecular structures of 2a, 2b, 2c, 3a, 3b and 3c were determined by X-ray crystallographic studies. These borolylated systems show intense blue/violet luminescence with Stokes shifts of 6200–9500 cm−1 and quantum yields of 0.33 to 0.98. The absorption maxima (296–351 nm) of these assemblies are reproduced well by TD-DFT computations (B3LYP/6–31G*), and arise from strong, low energy HOMO–LUMO transitions. From molecular orbital computations on optimised geometries of these diazaborolyl systems, the LUMO is located mainly on the thiophene/benzene bridge (66–92%) while the HOMO is largely benzodiazaborolyl in character (53–83%). The S1←S0 absorption bands are thus assigned to π(diazaborolyl)–π*(thiophene/benzene) transitions. Computations on related bis(boryl) dithiophenes [with diarylboryl e.g. Ph2B, Mes2B, (C6F5)2B and FMes2B (Mes = 2,4,6-Me3C6H2; FMes = 2,4,6-(CF3)3C6H2), dioxaborolyl and other diazaborolyl groups] reveal strong, low energy UV-visible absorption bands arising from π(thiophene)–π*(thiophene) transitions, with increasing boron participation in the LUMO of the diarylboryl and especially the highly fluorinated systems.

 Please wait while we load your content...
Please wait while we load your content...