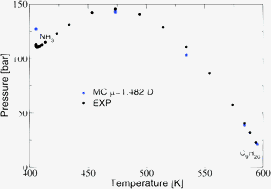
In this paper we investigate the phase diagram of pure dipolar substances and their mixtures with short alkanes, using grand canonical Monte Carlo simulations of simplified coarse-grained models. Recently, an efficient coarse-grained model for simple quadrupolar molecules, based on a Lennard-Jones (LJ) interaction plus a spherically averaged quadrupolar potential, has been shown to be successful in predicting single-component and mixture phase diagrams. Motivated by these results, we investigate the phase diagrams of simple dipolar molecules (and their mixtures with alkanes) using a spherically averaged potential. First, we test the model on pure components. A generalized (state-dependent) mapping procedure allows us to recycle Monte Carlo results of the simple Lennard-Jones (LJ) potential. Considering ammonia, nitrous oxide, and hydrogen sulfide, we generally observe improvements in the single-component phase diagram compared to a pure LJ description, but also some discrepancies in the coexistence pressure near the critical point and in the liquid branch of the coexistence densities well below criticality. In addition, we present results for mixtures. We consider mixtures of ammonia (NH3) with methane (CH4), nonane (C9H20) and hexadecane (C16H34)—for which experimental results are available—and compare the predictions from this modeling ansatz with predictions from simple LJ models. We also present results for the hydrogen sulfide–pentane mixture (H2S and C5H12) for which big discrepancies between simulations and experiments are present. Possible explanations for these discrepancies and limitations of the modeling are discussed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...