Study of substituent effects for aliphatic CH3–X compounds by resonant Auger spectroscopy
Abstract
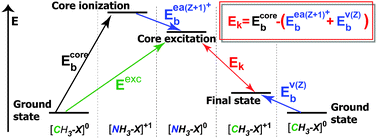
In this study linear free energy relationships (LFER) are applied to the relaxation of core-excited states in aliphatic CH3X compounds. There are at least two valence orbitals involved in Auger decay following CCH31s →σ*C–X excitation, which may as well be localized on the substituents in this series. Therefore, the observed property cannot be strictly attached to the so-called reaction center. The behavior of substituent constants in the description of core-hole processes in

 Please wait while we load your content...
Please wait while we load your content...