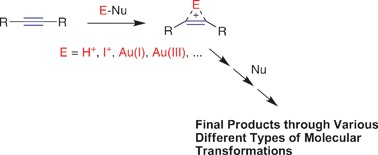
The reaction of 2-alkynyl-1-methylene azide aromatics 1 with iodine gives 1,3-disubstituted 4-iodoisoquinolines 3, and the treatment of 1 with a gold–silver combined catalyst affords isoquinolines 4. On the other hand, TfOH-catalyzed reaction of 1 produces 1,3-dipolar cycloadducts, triazoles 5. Computations reveal that “non-symmetrical” and “slightly non-symmetrical” coordinations between the triple bond and electrophiles (cationic Au and iodonium species) are prone to give the isoquinolines, while “symmetrical” coordination with electrophiles (a Brønsted acid and Au(I)) affords the triazoles. Keeping this background material in mind, the reactions through alkyne activation with electrophiles are surveyed. In most cases, products having similar structural frameworks were obtained through alkyne activation with Brønsted acids, iodine and gold complexes; the difference is whether H or I is incorporated in the final products. However, in a few cases, different reactivities and product structures were observed between those three reagents and catalysts.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...