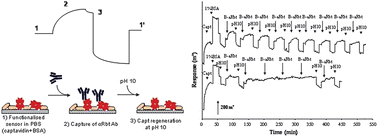
Functionalisation of a biosensor's sensing surface with the appropriate biorecognition elements is essential for the correct performance of the biosensor. In this paper, we investigate by Surface Plasmon Resonance (SPR) if captavidin, a recently described biotin-binding regenerable protein, could be used to bind and release biotinylated biocomponents for the development of regenerable biosensors. In our experiments, biotinylated antibodies were successfully subjected to up to nine serial capture–release events from the captavidin-functionalised surface. Up to three consecutive captures were possible when a protein target had been subsequently added. On the other hand, biotinylated bacteria were also efficiently captured and released from the gold surface, suggesting also the suitability of captavidin for the development of whole-cell regenerable biosensors. Our results indicate that captavidin is a promising regenerable molecular tool that could be used during biosensor optimisation and validation, and that captavidin-modified surfaces could be fine-tuned into truly reusable sensors.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...