A series of bis(E-dimesitylborylethenyl)-substituted arenes, namely arene = 1,4-benzene, 1,4-tetrafluorobenzene, 2,5-thiophene, 1,4-naphthalene, 9,10-anthracene, 4,4′-biphenyl, 2,7-fluorene, 4,4′-E-stilbene, 4,4′-tolan, 5,5′-(2,2′-bithiophene), 1,4-bis(4-phenylethynyl)benzene, 1,4-bis(4-phenylethynyl)tetrafluorobenzene and 5,5″-(2,2′:5′,2″-terthiophene), have been synthesised viahydroboration of the corresponding diethynylarenes with dimesitylborane. Their absorption and emission maxima, fluorescence lifetimes and quantum yields are reported along with the two-photon absorption (TPA) spectra and TPA cross-sections for the 5,5′-bis(E-dimesitylborylethenyl)-2,2′-bithiophene and 5,5′-bis(E-dimesitylborylethenyl)-2,2′:5′,2″-terthiophene derivatives. The TPA cross-section of the latter compound of ca. 1800 GM is the largest yet reported for a 3-coordinate boron compound and is in the range of the largest values measured for quadrupolar compounds with similar conjugation lengths. The X-ray crystal structures of 1,4-benzene, 2,5-thiophene, 4,4′-biphenyl and 5,5″-(2,2′:5′,2″-terthiophene) derivatives indicate π-conjugation along the BC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–arene–C
C–arene–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CB chain. Theoretical studies show that the second molecular hyperpolarisabilities, γ, in each series of compounds are generally related to the HOMO energy, which itself increases with increasing donor strength of the spacer. A strong enhancement of γ is predicted as the number of thiophene rings in the spacer increases.
CB chain. Theoretical studies show that the second molecular hyperpolarisabilities, γ, in each series of compounds are generally related to the HOMO energy, which itself increases with increasing donor strength of the spacer. A strong enhancement of γ is predicted as the number of thiophene rings in the spacer increases.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–
C–![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CB chain. Theoretical studies show that the second molecular hyperpolarisabilities, γ, in each series of compounds are generally related to the HOMO energy, which itself increases with increasing donor strength of the spacer. A strong enhancement of γ is predicted as the number of thiophene rings in the spacer increases.
CB chain. Theoretical studies show that the second molecular hyperpolarisabilities, γ, in each series of compounds are generally related to the HOMO energy, which itself increases with increasing donor strength of the spacer. A strong enhancement of γ is predicted as the number of thiophene rings in the spacer increases.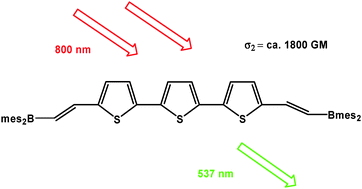

 Please wait while we load your content...
Please wait while we load your content...