Manipulation and charge determination of proteins in photopatterned solid supported bilayers†
Abstract
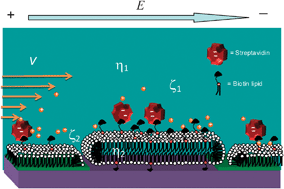
This work demonstrates the use of deep UV micropatterned
* Corresponding authors
a
School of Physics and Astronomy, University of Leeds, Leeds, UK
E-mail:
s.d.evans@leeds.ac.uk
Fax: +44 113 343 3900
Tel: +44 113 343 3852
b Self-Organising Molecular Systems (SOMS) Centre, University of Leeds, UK
This work demonstrates the use of deep UV micropatterned

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
