A variety of zero-valent metals (Al0, Cu0, Fe0, Mg0, Ni0, Si0 and Zn0), and Cu0 and Fe0 nano-sized particles were evaluated for reactivity towards the reduction of Cr(VI) in a surfactant preparation (Tween® 20) under a selection of reaction conditions. At circum neutral pHs, a rapid inactivation of the surface was observed for almost all of the tested metals and complete reduction of Cr(VI) was achieved at acidic pH only by using Cu0, Fe0, Mg0 or Zn0. A considerable increase in reactivity (up to 100-fold) was observed for reductive remediation with nano-sized particles. The use of a solid support as a dispersant and stabilizer circumvented the tendency of freshly prepared nano-particles to either react with surrounding media or agglomerate, resulting in the formation of much larger flocs and appreciable loss in reactivity. The formation of clusters by polymeric structures provided an extra protection of the nano-particle surface with a striking improvement in their reactivity mainly at more alkaline conditions. Therefore the incorporation of an innocuous stabilizer can substantially enhance the stability of nano-particles for environmental transformations.
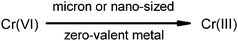
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
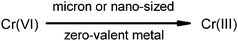

 Please wait while we load your content...
Please wait while we load your content...