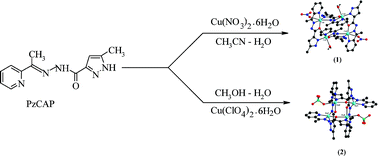
The pyrazole derived Schiff base polytopic ligand 5-methyl-N′-[1-(pyridin-2-yl)ethylidene]-1H-pyrazole-3-carbohydrazide (PzCAP), prepared by the reaction between 5-methylpyrazole-3-carbohydrazide and 2-acetyl pyridine, has two potentially bridging functional groups [μ-O and μ-(N-N)] and consequently can exhibit different coordination conformations. Two tetranuclear homoleptic copper(II) 2 × 2 rectangular grid-complexes [Cu4(PzCAP)4(NO3)2] (NO3)2·8H2O (1) and [Cu4(PzCAP)4(ClO4)2] (ClO4)2 (2) were formed by a strict self-assembly process employing metal and ligand under 1:1 mol proportion. Each pair of the ligand molecules in the two complexes are arranged in roughly parallel fashion but under different conformations. The ligand PzCAP contains terminal pyridine and pyrazole residues bound to a central flexible diazine subunit (N-N). The rectangular Cu(II) 2 × 2 grid complexes having [Cu4(μ-N-N)2(μ-O)2] core involve a mixture of two diazine (Cu-N-N-Cu ∼160°) and two alkoxo (Cu-O-Cu ∼138°) bridges along the length and breadth respectively. In the [Cu4(μ-N-N)2(μ-O)2] core in 1, out of the four Cu(II) centers, all are hexa-coordinated but there are two penta-coordinated and two hexa-coordinated Cu(II) centers in the same core of 2. Each complex having the central [Cu4(μ-N-N)2(μ-O)2] core, exhibits quite different magnetic interactions among the metal centers. The paramagnetic Cu(II) centers bridged through the diazine fragment are involved in anti-ferromagnetic interaction while a dominant ferromagnetic interaction prevails between the alkoxo-bridged Cu(II) centers. The [Cu4(μ-N-N)2(μ-O)2] cluster in 1 shows both ferromagnetic and anti-ferromagnetic interaction (J1 = −0.80 cm−1 and J2 = +3.49 cm−1), a very unusual characteristic in this system while the same cluster in 2 exhibits dominant anti-ferromagnetic coupling (J1 = −89.1 cm−1 and J2 = +5.5 cm−1) through the trans Cu-(N-N)-Cu bridging arrangement, typical for systems of this sort. Both the complexes 1 and 2 have been characterized structurally, magnetically and spectroscopically. The exchange pathways parameters (J1 and J2) have also been evaluated from density functional theoretical calculations to corroborate the bridging signatures with experimental findings.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...