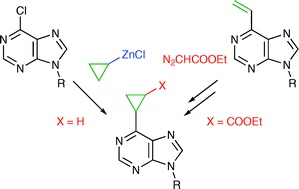
Synthesis of substituted 6-cyclopropylpurine bases and nucleosides by cross-coupling reactions or cyclopropanations†
Abstract
Synthesis of novel
- This article is part of the themed collection: Chemistry Nobel 2010 Web Collection: Cross-coupling reactions in organic chemistry

 Please wait while we load your content...
Please wait while we load your content...