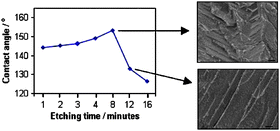
“Etch and coat” methods, potentially useful for large scale production of superhydrophobic surfaces on metals, have been extended by introduction of a thin 20 nm Au interlayer which allowed thiol surface modifiers to be investigated. Fe, Ti and Zn surfaces modified in this way with a polyfluorothiol generally had lower contact angles, θ, than their silane modified equivalents but were more uniform and reproducible. The higher θ values for silanes appear to be associated with an increased effective roughness that is not seen in the thiol coatings, which follow the underlying surface. For Fe, Ti and Zn, θ increased to a maximum with etching time but then decreased significantly. Electron microscopy of samples etched for long times showed fewer grain boundaries, suggesting that the loss of hydrophobicity was related to reduced roughness. This was confirmed through quantitative measurements of roughness parameters which were directly correlated with the observed contact angles through the Wenzel equation. The observation of this behaviour for 3 different metals suggests it is a general effect and the assumption that increased etch times will give increased roughness is incorrect. Although etching, Au coating and thiol modification did not provide sufficient roughness for superhydrophobicity, electrodeposition of a Cu layer onto the metal substrates followed by electroless deposition of silver gave uniform surfaces with very high contact angles (up to θ = 168.9°) and low roll-off angles (α < 1°). This method is sufficiently simple and inexpensive to be suitable for application on industrial scales, for example in marine or aeronautical engineering.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...