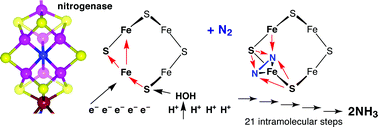
Using density functional calculations, a complete chemical mechanism has been developed for the reaction N2 + 6e− + 6H+→ 2NH3 catalyzed by the Fe7MoS9Nc(homocitrate) cofactor (FeMo-co) of the enzyme nitrogenase. The mechanism is based on previous descriptions of the generation of H atoms on FeMo-co by proton relay through a protein path terminating in water molecule 679, and preserves the model (which explains much biochemical data) for vectorial migration of H atoms to two S atoms and two Fe atoms of FeMo-co. After calculation of the energy profiles for the many possible sequences of steps in which these H atoms are transferred to N2 and its hydrogenated intermediates, a favourable pathway to 2NH3 was developed. Transition states and activation potential energies for the 21 step mechanism are presented, together with results for some alternative branches. The mechanism develops logically from the η2-coordination of N2 at the endo position of one Fe atom of prehydrogenated FeMo-co, consistent with the previous kinetic–mechanistic scheme of Thorneley and Lowe, and passes through bound N2H2 and N2H4 intermediates. This mechanism is different from others in the literature because it uses a single replenishable path for serial supply of protons which become H atoms on FeMo-co, migrating to become S–H and Fe–H donors to N2 and to the intermediates that follow. The new paradigm for the chemical catalysis is that hydrogenation of N2 and intermediates is intramolecular and does not involve direct protonation from surrounding residues which appear to be unable to provide a replenishable supply of 6H+. Many steps in this intramolecular hydrogenation are expected to be enhanced by H tunneling.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...