Molecular recognition in homogeneous transition metal catalysis: a biomimetic strategy for high selectivity
Abstract
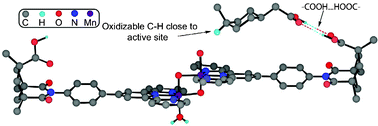
Traditional methods for selectivity control in homogeneous transition metal catalysis either employ steric effects in a binding pocket or chelate control. In a supramolecular strategy, encapsulation of the substrate can provide useful shape and size selectivity. A fully developed molecular recognition strategy involving hydrogen bonding or solvophobic forces has given almost completely regioselective functionalization of remote, unactivated C–H bonds.

 Please wait while we load your content...
Please wait while we load your content...