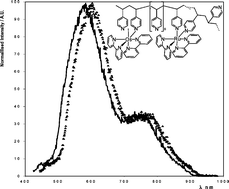
Luminescence quenching of the metallopolymers [Ru(bpy)2(PVP)10]2+ and [Ru(bpy)2(PVP)10Os(bpy)2]4+, both in solution and as thin films, is reported, where bpy is 2,2′-bipyridyl and PVP is poly(4-vinylpyridine). When the metallopolymer is dissolved in ethanol, quenching of the ruthenium excited state, Ru2+*, within [Ru(bpy)2(PVP)10]2+ by [Os(bpy)3]2+ proceeds by a dynamic quenching mechanism and the rate constant is (1.1 ± 0.1) × 1011 M−1 s−1. This quenching rate is nearly two orders of magnitude larger than that found for quenching of monomeric [Ru(bpy)3]2+ under the same conditions. This observation is interpreted in terms of an energy transfer quenching mechanism in which the high local concentration of ruthenium luminophores leads to a single [Os(bpy)3]2+ centre quenching the emission of several ruthenium luminophores. Amplifications of this kind will lead to the development of more sensitive sensors based on emission quenching. Quenching by both [Os(bpy)3]2+ and molecular oxygen is significantly reduced within a thin film of the metallopolymer. Significantly, in both optically driven emission and electrogenerated chemiluminescence, emission is observed from bothruthenium and osmium centres within [Ru(bpy)2(PVP)10Os(bpy)2]4+ films, i.e. the ruthenium emission is not quenched by the coordinated[Os(bpy)2]2+ units. This observation opens up new possibilities in multi-analyte sensing since each luminophore can be used to detect separate analytes, e.g.guanine and oxoguanine.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...