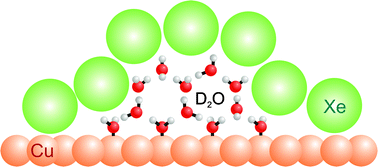
We investigate the binding site of solvated electrons in amorphous D2O clusters and D2O wetting layers adsorbed on Cu(111) by means of two-photon photoelectron (2PPE) spectroscopy. On the basis of different interactions of bulk- or surface-bound solvated electrons with rare gas atoms, titration experiments using Xe overlayers reveal the location of the electron solvation sites. In the case of flat clusters with a height of 2–4 bilayers adsorbed on Cu(111), solvated electrons are found to reside at the ice–vacuum interface, whereas a bulk character is found for solvated electrons in wetting layers. Furthermore, time-resolved experiments are performed to determine the origin of the transition between these different solvation sites with increasing D2O coverage. We employ an empirical model calculation to analyse the rate of electron transfer back to the substrate and the energetic stabilization of the solvated electrons, which allows further insight into the binding site for clusters. We find that the solvated electrons reside at the edges of the clusters. Therefore, we attribute the transition from surface- to bulk-solvation to the coalescence of the clusters to a closed ice film occurring at a nominal coverage of 2–3 BL, while the distance of the binding sites to the metal–ice interface is maintained.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...