The semiconductor catalyzed photoaddition of cyclopentene or cyclohexene to various novel electron-poor imines of type p-XC6H4(CN)C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N(COPh) (X = H, F, Cl, Br, Me, MeO) was investigated as a function of the nature of the cadmium sulfide photocatalyst. Irradiation (λ
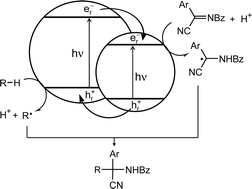
≥ 350 nm) of silica supported cadmium sulfide surprisingly did not afford the expected olefin–imine adducts but an imine hydrocyanation product via an unprecedented dark reaction. However, when silica was replaced by zinc sulfide as the support for cadmium sulfide, the expected homoallylic N-benzoyl-α-amino cyanides were isolated in yields of 65–84%. Thus, chemoselectivity is introduced through replacing an insulating by a semiconducting support, a hitherto unknown effect in semiconductor photocatalysis. From the sign of the time resolved photovoltage it is found that the mixed metal sulfide interface CdS/ZnS increases the lifetime of photogenerated electron–hole pairs by about one order of magnitude as compared to the SiO2/CdS system. The reaction rate increases with increasing imine σ-Hammett constants and decreasing stability of intermediate benzyl radicals.
N(COPh) (X = H, F, Cl, Br, Me, MeO) was investigated as a function of the nature of the cadmium sulfide photocatalyst. Irradiation (λ
≥ 350 nm) of silica supported cadmium sulfide surprisingly did not afford the expected olefin–imine adducts but an imine hydrocyanation product via an unprecedented dark reaction. However, when silica was replaced by zinc sulfide as the support for cadmium sulfide, the expected homoallylic N-benzoyl-α-amino cyanides were isolated in yields of 65–84%. Thus, chemoselectivity is introduced through replacing an insulating by a semiconducting support, a hitherto unknown effect in semiconductor photocatalysis. From the sign of the time resolved photovoltage it is found that the mixed metal sulfide interface CdS/ZnS increases the lifetime of photogenerated electron–hole pairs by about one order of magnitude as compared to the SiO2/CdS system. The reaction rate increases with increasing imine σ-Hammett constants and decreasing stability of intermediate benzyl radicals.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N(COPh) (X = H, F, Cl, Br, Me, MeO) was investigated as a function of the nature of the cadmium sulfide photocatalyst. Irradiation (λ
≥ 350 nm) of
N(COPh) (X = H, F, Cl, Br, Me, MeO) was investigated as a function of the nature of the cadmium sulfide photocatalyst. Irradiation (λ
≥ 350 nm) of 

 Please wait while we load your content...
Please wait while we load your content...