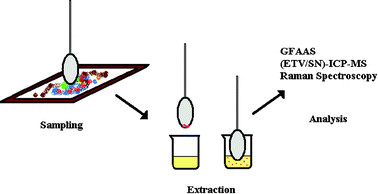
Inorganic pigments in paint have been used by artists throughout history, primarily because of their resistance to chemical change over long periods of time. Many of the historically-important pigments consist of transition metal oxides, sulfides or carbonates. The determination of metals in pigments is a powerful tool that can be used by conservators and researchers to identify pigments. In this study, pigments were sampled using a dry cotton bud that was contacted lightly with the surface of painted objects to remove a small quantity of pigment (<1 μg). This sampling approach causes no visible damage to a paint surface. Evidence of contact cannot be detected visually, even with the aid of a magnifying glass. Prior to analysis, pigment metals were extracted from the cotton with concentrated HNO3 in a 2 ml polystyrene beaker. The pigment metals were determined using graphite furnace atomic absorption spectroscopy (GFAAS) and electrothermal vaporization inductively coupled plasma mass spectrometry (ETV-ICP-MS). The small sample size requirement and high sensitivity of these instruments make them very suitable for analyzing pigment metals. Solution nebulization SN-ICP-MS was also used for obtaining a mass scan for most elements in the periodic table. The application of both GFAAS and ICP-MS is described for determining the identity of the metals in different pigments of two cultural objects (a painting and a map), which had darkened over time. Raman spectroscopy was used for confirming the identity of a darkened red pigment in a map as Red Lead (Pb3O4).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...