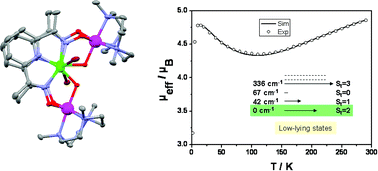
The reaction of [Cu(DapdoH2)2]2+ ions with [CrIIIL(MeOH)2Br]2+ ions (L = 1,4,7-trimethyl-1,4,7-triazacyclononane; DapdoH2 = 2,6-diacetylpyridine dioxime) produced in situ in methanol, yielded, in the presence of triethylamine, the complex [L2CrIII2(Dapdo)2(OH)2CuII2Br2](ClO4)21. It has been characterized on the basis of elemental analysis, IR spectroscopy and variable-temperature (2–298 K) magnetic susceptibility measurements. The molecular structure established by X-ray diffraction consists of a [CrIII2(µ-NO)4(µ-OH)2CuII2]4+-core, which can be considered as two edge-sharing triangular CrCu2-units. The variable-field variable-temperature magnetic measurements revealed a ground state of St = 2 with uncommon antiferromagnetic exchange interactions between the chromium(III) and copper(II) centers: JA = −79 ± 2 cm−1, JB = −17 ± 1.7 cm−1, where JA represents the interactions through a combination of oximate (>N–O-) and a hydroxo-bridging, while JB is the exchange through only a two-atom oximate (>N–O-) group.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...