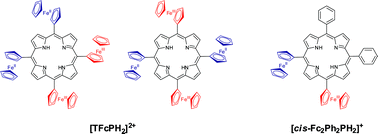
Metal-free 5,10,15,20-tetraferrocenylporphyrin and 5,10-bisferrocenyl-15,20-bisphenylporphyrin have been prepared and characterized by UV-Vis, MCD, 1H, 13C, and variable-temperature NMR, APCI- and ESI-MS, and Mössbauer spectroscopy, while their redox properties were investigated using electrochemical (cyclic voltammetry and differential pulse voltammetry), spectroelectrochemical, and chemical oxidation approaches. The electronic structure calculations at Density Functional Theory level reveal that both compounds adopt saddle conformations and the HOMOs in both complexes are predominantly metal-centered, while the LUMOs predominantly consist of porphyrin π* orbitals. In spite of the rotational freedom of ferrocenyl substituents at room temperature, both metal-free 5,10,15,20-tetraferrocenylporphyrin and 5,10-bisferrocenyl-15,20-bisphenylporphyrin are able to form mixed-valence states upon the successive ferrocene-based two- and one-electron oxidations, respectively, as confirmed by UV-Vis, MCD, Mössbauer, electro-, and spectroelectrochemical methods, and thus, the earlier suggested (Boyd et al. Chem. Commun., 1999, 637) requirements for the formation of mixed-valence states in ferrocene-containing porphyrins should be revised.

 Please wait while we load your content...
Please wait while we load your content...