Heterocycle synthesis by copper facilitated addition of heteroatoms to alkenes, alkynes and arenes
Abstract
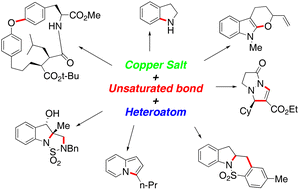
The de nouvo synthesis of small organic heterocyclic molecules has benefited from recent protocols for copper-facilitated additions of heteroatoms to
- This article is part of the themed collection: Organometallics in heterocyclic chemistry

 Please wait while we load your content...
Please wait while we load your content...