Photoinduced charge separation in self-assembled cofacial pentamers of zinc-5,10,15,20-tetrakis(perylenediimide)porphyrin†
Abstract
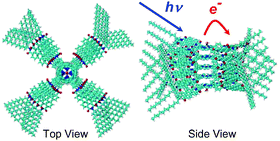
A bichromophoric electron donor–acceptor molecule composed of a zinc tetraphenylporphyrin (ZnTPP) surrounded by four perylene-3,4:9,10-bis(dicarboximide) (PDI) chromophores (ZnTPP-PDI4) was synthesized. The properties of this molecule were compared to a reference molecule having ZnTPP covalently bound to a single PDI (ZnTPP-PDI). In toluene, ZnTPP-PDI4 self-assembles into monodisperse aggregates of five molecules arranged in a columnar stack, (ZnTPP-PDI4)5. The monodisperse nature of this assembly contrasts sharply with previously reported ZnTPP-PDI4 derivatives having 1,7-bis(3,5-di-t-butylphenoxy) groups (ZnTPP-PPDI4). The size and structure of this assembly in solution was determined by small angle X-ray scattering (SAXS) using a high flux synchrotron X-ray source. The ZnTPP-PDI reference molecule does not aggregate. Femtosecond transient absorption spectroscopy shows that laser excitation of both ZnTPP-PDI and (ZnTPP-PDI4)5 results in quantitative formation of ZnTPP+˙-PDI−˙ radical ion pairs in a few picoseconds. The transient absorption spectra of (ZnTPP-PDI4)5 suggest that the PDI−˙ radicals interact strongly with adjacent PDI molecules within the columnar stack. Charge recombination occurs more slowly within (ZnTPP-PDI4)5 (τ = 4.8 ns) than it does in ZnTPP-PDI (τ = 3.0 ns) producing mostly ground state as well as a modest yield of the lowest triplet state of PDI (3*PDI). Formation of 3*PDI occurs by rapid spin–orbit induced intersystem crossing (SO-ISC) directly from the singlet radical ion pair as evidenced by the electron spin polarization pattern exhibited by its time-resolved electron paramagnetic resonance spectrum.

 Please wait while we load your content...
Please wait while we load your content...