Separable cooperative and localized translational motions of water confined by a chemically heterogeneous environment†
Abstract
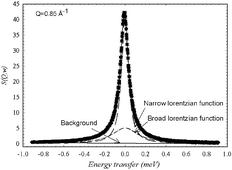
We report quasi-elastic neutron scattering experiments at two resolutions that probe timescales of picoseconds to nanoseconds for the hydration dynamics of

 Please wait while we load your content...
Please wait while we load your content...