Spectroscopy and potential energy surface of the H2–CO2 van der Waals complex: experimental and theoretical studies†‡
Abstract
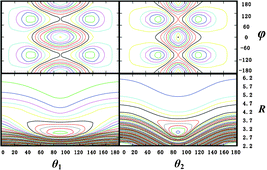
A 4-D ab initio potential energy surface is calculated for the intermolecular interaction of hydrogen and carbon dioxide, using the CCSD(T) method with a large basis set. The surface has a global minimum with a well depth of 212 cm−1 and an intermolecular distance of 2.98 Å for a planar configuration with both the O–C–O and H–H axes perpendicular to the intermolecular axis. Bound state calculations are performed for the H2–CO2 van der Waals complex with H2 in both the para and ortho spin states, and the binding energy of paraH2–CO2
(50.4 cm−1) is found to be significantly less than that of orthoH2–CO2
(71.7 cm−1). The surface supports 7 bound intermolecular vibrational states for paraH2–CO2 and 19 for orthoH2–CO2, and the lower rotational levels with J
≤ 4 follow an asymmetric rotor pattern. The calculated

 Please wait while we load your content...
Please wait while we load your content...