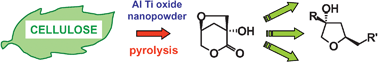
As a bulk component of plant biomass, cellulose plays a key role in the route leading to viable chemicals from renewable resources. Pyrolysis is a thermal treatment that converts cellulose into a liquid material (bio-oil) containing dehydrated monomers, among which the anhydrosugars levoglucosan (LGA) and levoglucosenone (LGO) have been widely investigated as chiral multifunctional synthons. A chiral cyclic hydroxylactone (LAC, 1) is also produced, but its potential as chemical intermediate has never been scrutinised, probably because it is generated only in minute amounts. In this study, experiments with a fixed bed reactor showed that the yields of LAC could be significantly increased by pyrolysing cellulose for few minutes at 350 °C in the presence of nanopowder (NP) titanium dioxide, aluminium oxide, and aluminium titanate (AlTi). When pyrolysis was conducted with NP AlTi at 500 °C, LAC became the principal anhydromonosaccharide and could be isolated from the resulting bio-oil with simple operations (6% overall yield). Its structure could be definitively assigned to the (1R,5S)-1-hydroxy-3,6-dioxabicyclo[3.2.1]octan-2-one by extensive NMR analysis and high resolution EI-MS experiments, providing a firm evidence of its previous attribution. The significance of LAC as building block for follow-up bioproducts was evaluated in some examples, including the synthesis of a suitable amide by the effective and eco-friendly microwave assisted methodology.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...