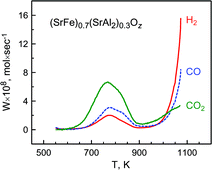
The steady-state CH4 conversion by oxygen permeating through mixed-conducting (SrFe)0.7(SrAl2)0.3Oz composite membranes, comprising strontium-deficient SrFe(Al)O3−δ perovskite and monoclinic SrAl2O4-based phases, occurs via different mechanisms in comparison to the dry methane interaction with the lattice oxygen. The catalytic behavior of powdered (SrFe)0.7(SrAl2)0.3Oz, studied by temperature-programmed reduction in dry CH4 at 523–1073 K, is governed by the level of oxygen nonstoichiometry in the crystal lattice of the perovskite component and is qualitatively similar to that of other perovskite-related ferrites, such as Sr0.7La0.3Fe0.8Al0.2O3−δ. While extensive oxygen release from the ferrite lattice at 700–900 K leads to predominant total oxidation of methane, significant selectivity to synthesis gas formation, with H2/CO ratios close to 2, is observed above 1000 K, when a critical value of oxygen deficiency is achieved. The steady-state oxidation over dense membranes at 1123–1223 K results, however, in prevailing total combustion, particularly due to excessive oxygen chemical potential at the membrane surface. In combination with surface-limited oxygen permeability, mass transport limitations in a porous layer at the membrane permeate side prevent reduction and enable stable operation of (SrFe)0.7(SrAl2)0.3Oz membranes under air/methane gradient. Taking into account the catalytic activity of SrFeO3−δ-based phases for the partial oxidation of methane to synthesis gas and the important role of mass transport-related effects, one promising approach for membrane development is the fabrication of thick layer of porous ferrite-based catalyst at the surface of dense (SrFe)0.7(SrAl2)0.3Oz composite.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...