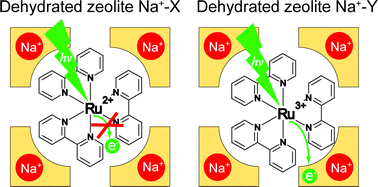
We carried out time-resolved luminescence and transient absorption studies of tris(2,2′-bipyridine)ruthenium(II) complex, Ru(bpy)32+ assembled in the supercages of zeolites X and Y exchanged with various alkali metal cations. The average lifetime of the luminescence decay, a measure of the photoinduced electron transfer (PET) rate, of Ru(bpy)32+* was found to decrease with increasing the electron-acceptor strength of the host which is represented by the Sanderson's electronegativity scale. This result strongly suggests that the zeolite host plays the role of electron acceptor for Ru(bpy)32+*. However, we could not detect Ru(bpy)33+ in the transient absorption spectra, most likely due to very low absorption coefficient of Ru(bpy)33+ and to the low efficiency of net PET. For the above observation to be made, it is essential to employ the dehydrated zeolite hosts to allow direct interaction between the guest Ru(II) complex and the host framework. The present study demonstrates the active role of the zeolite hosts during the PET of incorporated Ru(bpy)32+ under the carefully controlled experimental conditions. This report demonstrates the fact that the zeolite hosts can serve as electron acceptors although in the past zeolites were shown to play the role of electron donors.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...