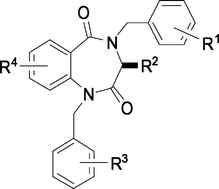
The expedient synthesis of various 1,4-benzodiazepine-2,5-dione compounds, particularly those having substituents at the C3-, N1- and N4-positions is achieved. The important features in these synthetic strategies include: (i) using the coupling reaction of isatoic anhydride with α-amino ester for direct construction of the core structure of 1,4-benzodiazepine-2,5-dione; (ii) using potassium carbonate as the base of choice for selective alkylation at the N1-site, while using lithiated 2-ethylacetanilide as the required base to furnish the N4-alkylation; and (iii) using 2-nitrobenzoyl chloride as a synthetic equivalent of anthranilic acid to facilitate the polyethylene resin-bound liquid-phase combinatorial synthesis. The prepared 1,4-benzodiazepine-2,5-dione compounds are evaluated for endothelin receptor antagonism by a functional assay that measures the inhibitory activity against the change of intramolecular calcium ion concentration induced by endothelin-1. The preliminary results indicate that 1,4-benzodiazepine-2,5-diones bearing two flanked aryl substituents at the N1- and N4-sites show better inhibitory activity than the corresponding unalkylated and N-monoalkylated compounds. A promising candidate, 1-benzyl-7-chloro-3-isopropyl-4-(3-methoxybenzyl)-1,4-benzodiazepine-2,5-dione (17b), exhibits an IC50 value in low nM range.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...