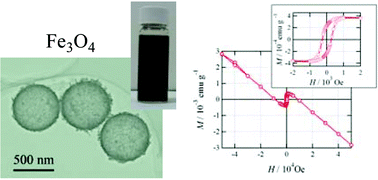
We carried out the phase selective preparations of hollow spheres of ccp- and hcp-Co, Co3O4, α-Fe, Fe3O4 and α-Fe2O3 with a diameter of ca. 600 nm and a shell thickness of 40 nm, using polystyrene (PS)-bead templates. The 600 nm PS beads were uniformly coated with cobalt or iron hydroxides by means of a homogeneous precipitation method. These cobalt-salt/PS hybrid particles were calcined in air at 400 °C and were transformed into Co3O4 hollow spheres. The hcp-Co hollow spheres were obtained by a reduction reaction of the Co3O4 particles under a stream of a 1 ∶ 1 mixed gas of H2 and N2 at 400 °C; in contrast, the ccp-Co hollow spheres were obtained by the calcination of the parent cobalt-salt/PS particles under H2–N2 at 400 °C. Hollow spheres of Fe3O4 and α-Fe2O3 were selectively prepared by calcination of iron-salt/PS particles at 400–500 °C in two different atmospheres, namely under H2–N2 and in air, respectively. Furthermore, the α-Fe2O3 particles were reduced into α-Fe under H2–N2 at 350 °C without any changes in morphology. These spheres were characterized by scanning electron microscopy (SEM), transmission electron microscopy (TEM), and X-ray diffraction (XRD) analyses. Magnetic measurements revealed a significant temperature dependence of the coercive field for the Fe3O4 (magnetite) particles, whereas the magnetic properties of the other particles were generally similar to those of the corresponding bulk samples. Surface chemical modifications were carried out on the Fe3O4 hollow spheres; the surfaces were grafted with poly(4-N-methylvinylpyridinium) brushes. This treatment was found to induce good dispersibility in water.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...