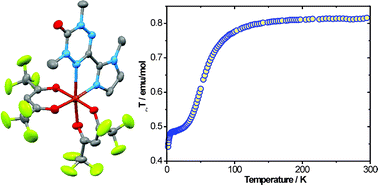
The synthesis, structures, and magnetic properties of several Cu(II) complexes of verdazyl radicals are presented. Reactions of chelating verdazyl radicals with either CuCl2·2H2O or Cu(hfac)2·2H2O produced 1 ∶ 1 Cu ∶ verdazyl complexes with either chloride or hfac ancillary ligands. Structural characterization reveals that the CuCl2 complexes of N,N′-dimethyl-3-(2-pyridyl)-6-oxoverdazyl or N,N′-bis(isopropyl)-3-(2-pyridyl)-6-oxoverdazyl have pseudo-square pyramidal copper ions with verdazyl rings bound in equatorial positions, while the Cu(hfac)2 complex of N,N′-dimethyl-3-(N-methyl-2-imidazolyl)-6-oxoverdazyl is Jahn–Teller distorted pseudo-octahedral and has the verdazyl nitrogen axially bound. Variable temperature magnetic susceptibility studies reveal that equatorially bound verdazyls are strongly antiferromagnetically coupled, while the axially bound radicals are weakly ferromagnetically coupled. Intermolecular magnetic interactions are also an important component of the overall magnetism in these systems.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...