Highly regioselective ring-opening of epoxides with thiophenols in ionic liquids without the use of any catalyst†
Abstract
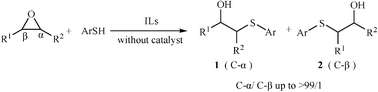
Clean thiolysis reaction of various epoxides in ionic liquids (ILs) without the use of any catalyst generates β-hydroxy sulfides in excellent yields (83–98%) with high regioselectivity and chemoselectivity under mild reaction conditions.

 Please wait while we load your content...
Please wait while we load your content...