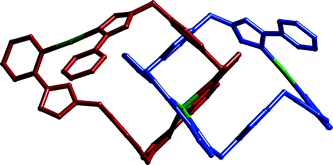
The coordination chemistry of a series of di- and tri-nucleating ligands with Ag(I), Hg(I) and Hg(II) has been investigated. Most of the ligands contain two or three N,N′-bidentate chelating pyrazolyl-pyridine units pendant from a central aromatic spacer; one contains three binding sites (2 + 3 + 2-dentate) in a linear sequence. A series of thirteen complexes has been structurally characterised displaying a wide range of structural types. Bis-bidentate bridging ligands react with Ag(I) to give complexes in which Ag(I) is four-coordinate from two bidentate donors, but the complexes can take the form of one-dimensional coordination polymers, or dinuclear complexes (mesocate or helicate). A tris-bidentate triangular ligand forms a complicated two-dimensional coordination network with Ag(I) in which Ag⋯Ag contacts, as well as metal–ligand coordination bonds, play a significant role. Three dinuclear Hg(I) complexes were isolated which contain an {Hg2}2+ metal–metal bonded core bound to a single bis-bidentate ligand which can span both metal ions. Also characterised were a series of Hg(II) complexes comprising a simple mononuclear four-coordinate Hg(II) complex, a tetrahedral HgII4 cage which incorporates a counter-ion in its central cavity, a trinuclear double helicate, and a trinuclear catenated structure in which two long ligands have spontaneously formed interlocked metallomacrocyclic rings thanks to cyclometallation of two of the Hg(II) centres.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...