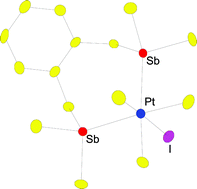
The planar Pt(II) monomers [PtMe2(L–L)] and [(PtMe2)2(L′–L′)2] dimers (L–L = R2Sb(CH2)3SbR2, o-C6H4(CH2SbMe2)2; L′–L′ = R2SbCH2SbR2; R = Me or Ph) are obtained in good yield via reaction of [PtMe2(SMe2)2] with L–L or L′–L′ in benzene. The Pt(IV) stibines, [PtMe3(L–L)I] (L–L = R2Sb(CH2)3SbR2, o-C6H4(CH2SbMe2)2 or 2 × SbPh3, SbMePh2 or SbMe2Ph) are obtained by treatment of [PtMe3I] with L–L in chloroform. These represent the first series of stable Pt(IV) stibine complexes. All of the products have been characterised by 1H, 13C{1H}, 195Pt NMR spectroscopy, electrospray mass spectrometry and analysis. Crystal structure determinations on [PtMe3{R2Sb(CH2)3SbR2}I], [PtMe3{o-C6H4(CH2SbMe2)2}I] and [PtMe3(SbPh3)2I] confirm the distorted octahedral environment at Pt, with fac Me groups and mutually cis Sb donor atoms. The Sb–Pt–Sb angle in the seven-membered chelate ring of the o-C6H4(CH2SbMe2)2 complex is ca. 96°, compared to <90° in the complexes with six-membered chelates. The C1-distibines R2SbCH2SbR2 afford only the dinuclear [(PtMe3)2(μ-R2SbCH2SbR2)(μ-I)2] in which the stibine ligand and two I atoms bridge two Pt atoms giving an edge sharing bioctahedral geometry which has been confirmed by a crystal structure analysis. The Pt(II) species undergo oxidative addition with MeI to give the corresponding Pt(IV) species, while the Pt(IV) species reductively eliminate ethane upon thermolysis.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...