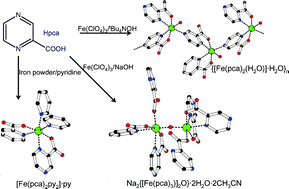
The synthesis and characterization of two new iron(II) complexes, [Fe(pca)2(py)2]·py (1) and {[Fe(pca)2(H2O)]·H2O}n (2) and one new iron(III) complex, Na2{[Fe(pca)3)]2O}·2H2O·2CH3CN (3) (pca− stands for 2-pyrazinecarboxylate), are reported. Complex 1 is obtained from the reaction of iron powder with 2-pyrazinecarboxylic acid. The reaction of Fe(ClO4)3·10H2O with Hpca in the presence of 3 equiv. of Bu4NOH yields 2, whereas the presence of NaOH yields 3. The molecular structure of 1 contains an iron(II) ion with a pseudo-octahedral environment resulting from the coordination of two pca− ligands in a bidentate chelating fashion and two pyridine molecules; π–π stacking interactions between pyridine and pyrazine rings lead to a one-dimensional chain. Complex 2 is an iron(II) coordination polymer with an infinite zig–zag motif and an Fe⋯Fe separation of 7.1 Å. In 2, the π–π stacking interactions involving the pyrazine rings and the strong hydrogen bonds between the coordinated water molecule and the carboxylate oxygens of two pca− ligands result in a three-dimensional network structure. Complex 3 consists of an anionic µ-oxo-bridged diiron(III) core with two crystallographically distinct iron(III) ions; the negative charge is compensated by two sodium cations. Complex 3 is assembled in a three dimensional network structure through coordination of Na(I) and hydrogen bond interactions. Temperature dependent magnetic susceptibility and Mössbauer spectroscopic studies indicate that 1 and 2 have similar magnetic properties. Both complexes are paramagnetic above 12 K, whereas antiferromagnetic ordering is observed below 12 K. The magnetic properties of 3 reveal strong intramolecular antiferromagnetic interactions between the two iron(III) ions with a J value of −221 cm−1; no long range intermolecular magnetic coupling is observed between 295 and 4.2 K.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...