Exploration of the potential energy surface of C4H4 for rearrangement and decomposition reactions of vinylacetylene: A computational study. Part I†‡
Abstract
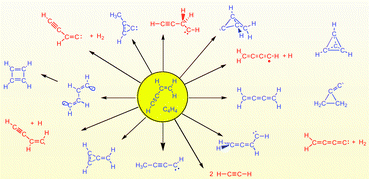
The potential energy surface (PES) of C4H4 was explored using quantum chemical methods (DFT, MP2, MP4, GVB-MP2, CCSD(T), G2M, CBSQ/APNO) and 43 different structures located at global and local minima were identified. The majority of these structures correspond to carbenes, a minority to closed shell systems and biradicals (carbyne structures were not investigated). Whereas the chemistry of the closed shell systems such as vinylacetylene (1), butatriene (2), methylenecyclopropene (3), cyclobutadiene (5) or tetrahedrane (15) is well known, the carbenes represent unusual structural entities. 2-Methyl-cycloprop-2-en-1-ylidene (4)
(ΔΔH(298)
= 36.2 kcal mol−1 relative to 1) in its σ2π0 electron configuration at the carbene C of the 1A ground state is of comparable stability to cyclobutadiene (5)
(ΔΔH(298)
= 33.4 kcal mol−1; exp. value: 32.1 kcal mol−1) as a result of aromatic 2π-delocalization; carbene 3-vinylidenecyclopropene (13)
(ΔΔH(298)
= 53.9 kcal mol−1) does not possess C2v symmetry but has the vinylidene group bent toward the three-membered ring (Cs-symmetry) thus representing a frozen path point of the chelotropic addition of :C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C: to ethene. Allenyl carbene (14) has a triplet ground state and two low lying excited singlet states of closed shell (2.5 kcal mol−1 higher) and open shell character (14.1 kcal mol−1). Carbene 14 is a crossing point on the C4H4 PES connecting closed-shell systems with each other. Because of the stability of 1, its rearrangement reactions are all connected with high activation enthalpies requiring 66 up to 92 kcal mol−1 so that they energetically overlap with the activation enthalpies typical of decomposition reactions (from 90 kcal mol−1 upward). The possible rearrangement reactions of 1 are investigated with a view to their relevance for the chemical behavior of the molecule under the conditions of Titan’s atmosphere.
C: to ethene. Allenyl carbene (14) has a triplet ground state and two low lying excited singlet states of closed shell (2.5 kcal mol−1 higher) and open shell character (14.1 kcal mol−1). Carbene 14 is a crossing point on the C4H4 PES connecting closed-shell systems with each other. Because of the stability of 1, its rearrangement reactions are all connected with high activation enthalpies requiring 66 up to 92 kcal mol−1 so that they energetically overlap with the activation enthalpies typical of decomposition reactions (from 90 kcal mol−1 upward). The possible rearrangement reactions of 1 are investigated with a view to their relevance for the chemical behavior of the molecule under the conditions of Titan’s atmosphere.

 Please wait while we load your content...
Please wait while we load your content...